What are Blood Cancers?
More than 1.5 million Americans are living with blood cancer — malignancies of the blood and bone marrow and other closely related disorders [1]. There are three main types of blood cancer: leukemia, lymphoma, and myeloma. In 2021, it is estimated that there were 90,390 new cases of lymphoma; 61,090 new cases of leukemia; and 34,920 new cases of myeloma (for a grand total of 186,400 new cases of blood cancer). Nearly 58,000 Americans will die from blood cancer in 2021 [2]. As is the case with nearly all types of cancer, blood cancers are more common among men than women. Additionally, blood cancer can strike at nearly any age. Leukemia (specifically, acute lymphoblastic leukemia) is the most common cancer in children.
Before going into further detail about these three types of blood cancers, let’s pause here to first review how blood cells are derived in the body.
Hematopoiesis: the Formation of Blood Cells
All components of the blood — red blood cells, immune (white blood) cells, and platelets — ultimately derive from a hemocytoblast, a multipotent stem cell located in red bone marrow (in the center of most bones). The process of hematopoiesis begins when a hematopoietic stem cell (hemocytoblast) divides into two new cells: one remains a hemocytoblast and the other begins a process of differentiation. (See image below).
The first “choice” is for the differentiated cell to become either a common myeloid progenitor or a common lymphoid progenitor. These progenitor cells are also stem cells, albeit slightly more specialized than the hemocytoblast from which they were derived. Similarly, when these cells divide, one remains a progenitor and the other becomes yet more specialized. This process continues, culminating in the production of mature and fully specialized cells — namely, red blood cells, immune cells, and platelets.
The common myeloid progenitor gives rise to the red blood cells, most of the innate immune cells (mast cells, basophils, neutrophils, eosinophils, and monocytes), and platelets. The common lymphoid progenitor: gives rise to the adaptive immune cells (T cells and B cells) and natural killer cells. Hematopoiesis inherently involves a lot of cell division. Therefore, there are many opportunities for mutations to occur and, hence, for cancer to arise. Cancer can emerge at just about any step during hematopoiesis (see the diagram below).
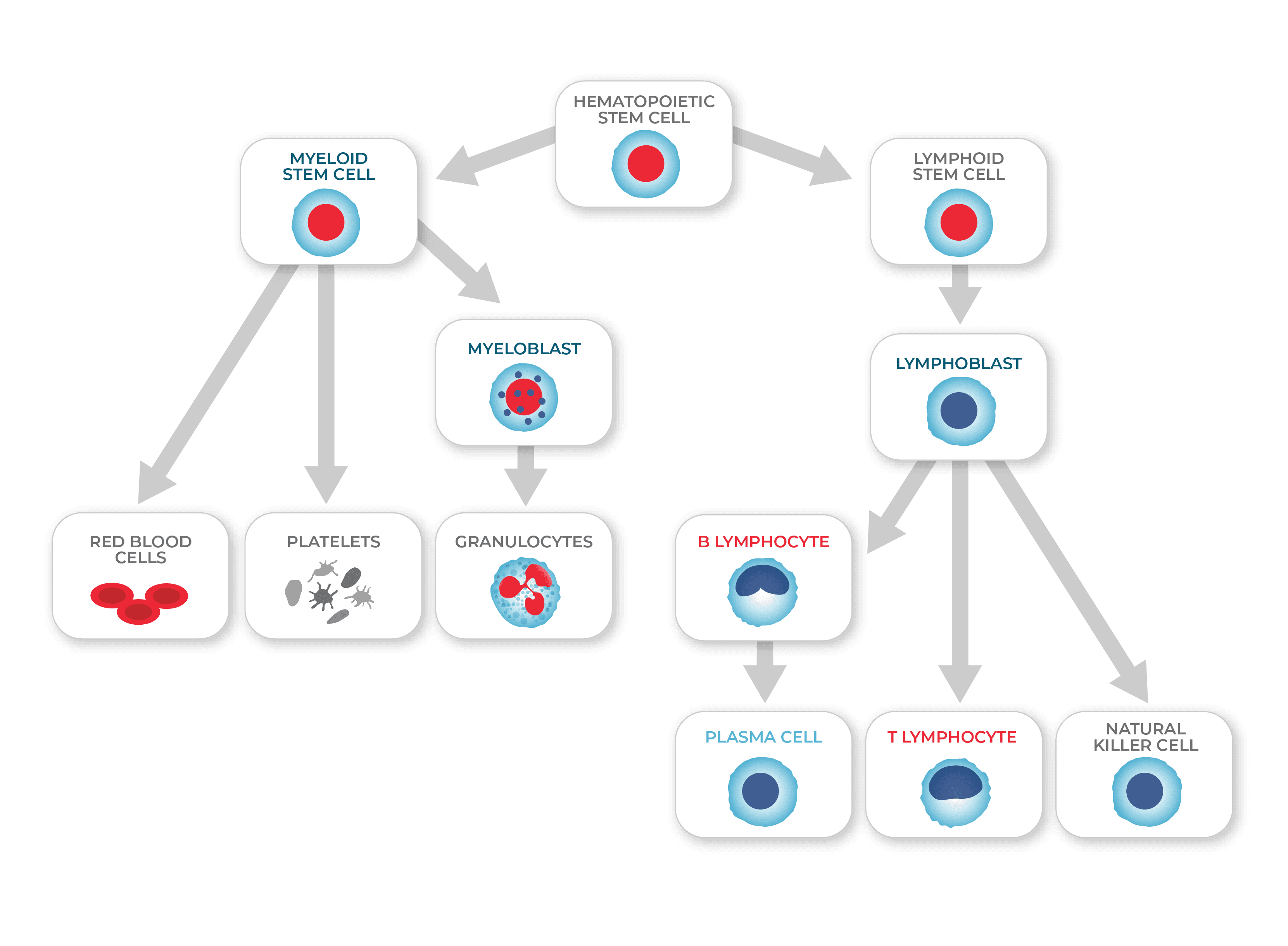
The process of hematopoiesis
Three Important Blood Cancers: Leukemia, Lymphoma, Myeloma
1. Leukemia
Leukemia is a cancer of the blood and bone marrow. Typically, a cancer stem cell arises in the bone marrow and malignant cells are eventually sent into the bloodstream. Leukemia is classified as either “acute” or “chronic,” depending on which type of cell has become cancerous:
Acute leukemias: these cancers emerge in stem cells, and malignant cells eventually take over the bone marrow and prevent the development of other blood cells (namely, red blood cells, immune cells, and platelets). As a result, acute leukemias can have dramatic onset with severe symptoms such as anemia, unusual bleeding, and frequent infections. Acute myeloid leukemia (AML), which derives from myeloid progenitor cells, mainly occurs in older adults, and acute lymphoblastic leukemia (ALL), which often derives from B cells, is the most common type of leukemia in children.
Chronic leukemias: derive from cells that are more differentiated. Patients with chronic leukemia are older adults who may be asymptomatic for years. For example, chronic myeloid leukemia (CML) arises from differentiated cells in the myeloid lineage and often leads to an overproduction of neutrophils. The prognosis of CML patients has improved dramatically due to new treatments [3]. Chronic lymphocytic leukemia (CLL), another chronic leukemia, is characterized by the improper proliferation of B cells.
2. Lymphoma
This cancer or the lymphatic system (tissues that combat disease) manifests as solid tumors found in lymphatic tissues, such as lymph nodes. Lymphomas are most often derived from T cells, B cells, or their precursors. B cell-derived lymphomas are the most common in the West.
Lymphomas can be broadly classified as either Hodgkin’s lymphoma or Non-Hodgkin’s lymphoma:
- Hodgkin’s lymphoma (HL): is a cancer of mature B cells, and it is identified by the presence of very unusual, multinucleated cells called Reed-Sternberg cells.
- Non-Hodgkin’s lymphoma (NHL): has a greater than 85% cure rate. NHL derives from B cells, T cells, or NK cells. The natural history of NHL is highly variable, with some cancers being aggressive and others indolent.
3. Myeloma
Myelomas are cancers of plasma cells (i.e., the mature, antibody-secreting version of B cells) and primarily mainly afflict older individuals. The disease is typically referred to as multiple myeloma because the cancer is usually detected in several locations. The myelomas emerge within bone marrow and can actually damage bones, resulting in bone pain. Patients with MM are susceptible to infections and kidney failure.
Treatments for Blood Cancer
There is no single treatment for blood cancer. A patient’s treatment is determined by his or her specific diagnosis.
Chemotherapy “Chemo” is a type of anti-cancer therapy that kills cancer cells by damaging their DNA or precluding cell division. The drugs target all fast-growing cells in the body, including cancer cells but some types of healthy cells as well. However, chemotherapy has nasty side effects because the drugs also kill healthy cells. Patients receiving chem are also more prone to infection because white blood cells are destroyed.
Bone Marrow Transplant For some blood cancers, another treatment option is a bone marrow transplant. Patients first must have their cancerous blood cells (located in the bone marrow) destroyed via chemotherapy or radiation. After this, the patient receives healthy hematopoietic stem cells, which can come from either the patient him/herself or from a donor.
Targeted Therapies Targeted therapies involve the use of drugs that target specific genes or proteins that are critical to the survival or propagation of cancer cells. Using these therapies requires precise knowledge of the molecular mechanisms underlying a patient’s cancer. For instance, a successful treatment for chronic myeloid leukemia (CML) uses a drug called imatinib to block a form of the enzyme tyrosine kinase that has a specific mutation that causes it to drive cell growth.10 One major drawback of targeted therapies is that cancers often evolve resistance to them. Thus, many oncologists are now testing out combination therapies to improve outcomes.
Looking Ahead: How Single-cell Research is Improving Therapies
Although there are several challenges to battling blood cancers, substantial progress has been made over the past few years. The advent of single-cell DNA sequencing has bolstered these efforts by allowing researchers to better understand the genetics of tumors. Although tumors are derived from a single cell gone “bad,” different cells acquire different sets of mutations as it grows. As this process repeats, several different subpopulations of cancer cells (called subclones) exist side-by-side in the same tumor — making the tumor genetically heterogeneous.
One way to understand tumor heterogeneity and evolution is by doing research on individual cancer cells. By sequencing the DNA of several cells extracted from a tumor, a “family tree” can be constructed of how a tumor evolved. Specifically, such information can allow researchers to determine what mutations must occur and in what order for a cell to become cancerous or for a subclone to become dominant. This knowledge can inform treatment strategies, particularly as we transition into an era of personalized medicine.
Single-cell analysis can also reveal how tumors become resistant to treatments. Because this approach can identify rare cell populations, it can detect even small frequencies of malignant cells that are resistant to chemotherapeutic drugs or other therapies. This information can be used in the clinic to inform treatment strategies that greatly reduce the risk of relapse. Equipped with single-cell DNA sequencing and other innovative technologies, oncologists and researchers are working hard to bring better treatment to blood cancer patients.
References




